polyantha
Well-Known Member
Hi everyone! I measured the photosynthesis performance of one of my Paph plants (philippinense x praestans) under different light tubes. The experiment setup:

A sealed glass box contains the plant and a CO2 meter. The collected data are sent to a data collector program on the computer. The CO2 is measured and one data set is saved per minute.
The idea is easy: The plant uses the light emitted from the light tubes to convert the light energy into usable chemical energy (produce sugar). The plant needs CO2 to do so and lets 02 go, so the air inside the box will contain fewer and fewer CO2 while the amount of 02 rises. The faster this happens the more efficient the photosynthesis is under those light conditions.

The curve above showing the result of a five hour experiment with standard daylight tubes. The plant reacts after 15 minutes after turning the lights on and starts doing photosynthesis. The CO2 concentration decreases very fast at the beginning, but it gets more and more hard for the plant to get the CO2 when the concentration gets very low, so the curve begins to flatten at the end.
I tried different light tubes, to find out which ones will be the best. I tried normal daylight, blue light (normally used in reef aquariums) and the well known purple plant light. Take a look at the curves:

The purple plant light was the most efficient of these three. The light spectrum of these light tubes is perfect for the plants, because they emit red and blue light at the absorption maximum of chlorophyll.
Many people doubt the efficiency of the Fluora tubes from Osram, but for my Paph plant is was clearly the best tube, followed by the blue light tube emitting a maximum at 440nm.
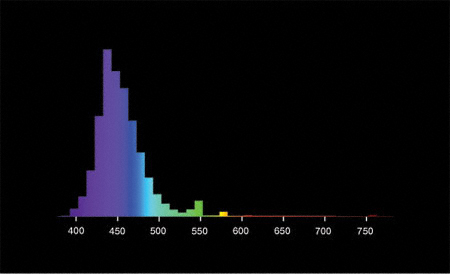
I repeated the experiment with T5 tubes (2 x 80W) and the blue light was better than the daylight tube again. I do not have purple tubes at the moment, but I will try those in the future (Fuji Purple T5). I guess they will be the most efficient again (check the emission curve).

I changed my lighting system some months ago and I noticed a much imporved growth on all my plants. At the moment I use T5 80W tubes 50% of the blue type and 50% of the daylight type. I will buy the Fuji Purple T5's and use them for 50%, blue 25% and daylight 25%. The problem is that those T5 purple light tubes are pretty expensive here in Switzerland

A sealed glass box contains the plant and a CO2 meter. The collected data are sent to a data collector program on the computer. The CO2 is measured and one data set is saved per minute.
The idea is easy: The plant uses the light emitted from the light tubes to convert the light energy into usable chemical energy (produce sugar). The plant needs CO2 to do so and lets 02 go, so the air inside the box will contain fewer and fewer CO2 while the amount of 02 rises. The faster this happens the more efficient the photosynthesis is under those light conditions.

The curve above showing the result of a five hour experiment with standard daylight tubes. The plant reacts after 15 minutes after turning the lights on and starts doing photosynthesis. The CO2 concentration decreases very fast at the beginning, but it gets more and more hard for the plant to get the CO2 when the concentration gets very low, so the curve begins to flatten at the end.
I tried different light tubes, to find out which ones will be the best. I tried normal daylight, blue light (normally used in reef aquariums) and the well known purple plant light. Take a look at the curves:

The purple plant light was the most efficient of these three. The light spectrum of these light tubes is perfect for the plants, because they emit red and blue light at the absorption maximum of chlorophyll.
Many people doubt the efficiency of the Fluora tubes from Osram, but for my Paph plant is was clearly the best tube, followed by the blue light tube emitting a maximum at 440nm.
I repeated the experiment with T5 tubes (2 x 80W) and the blue light was better than the daylight tube again. I do not have purple tubes at the moment, but I will try those in the future (Fuji Purple T5). I guess they will be the most efficient again (check the emission curve).

I changed my lighting system some months ago and I noticed a much imporved growth on all my plants. At the moment I use T5 80W tubes 50% of the blue type and 50% of the daylight type. I will buy the Fuji Purple T5's and use them for 50%, blue 25% and daylight 25%. The problem is that those T5 purple light tubes are pretty expensive here in Switzerland